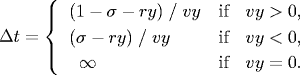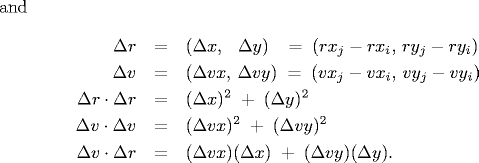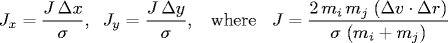COS 226 Programming Assignment
Molecular Dynamics Simulation of Hard Spheres
Simulate the motion of N colliding particles according to
the laws of elastic collision using event-driven simulation.
Such simulations are widely used in molecular dynamics (MD)
to understand and predict properties of physical systems at
the particle level.
This includes the motion of molecules in a gas,
the dynamics of chemical reactions, atomic diffusion,
sphere packing, the stability of the rings around Saturn,
the phase transitions of cerium and cesium,
one-dimensional self-gravitating systems, and front propagation.
The same techniques apply to other domains that involve physical
modeling of particle systems including computer graphics,
computer games, and robotics.
Hard sphere model.
The hard sphere model (billiard ball model) is an
idealized model of the motion of atoms or molecules in a container.
In this assignment, we will focus on the two-dimensional version
called the hard disc model. The salient properties of this
model are listed below.
- N particles in motion, confined in the unit box.
- Particle i has known
position (rxi, ryi),
velocity (vxi, vyi),
mass mi,
and radius σi.
- Particles interact via elastic collisions with
each other and with the reflecting boundary.
- No other forces are exerted. Thus, particles travel
in straight lines at constant speed between collisions.
This simple model plays a central role in statistical mechanics, a field
which relates macroscopic
observables (e.g., temperature, pressure, diffusion constant) to microscopic
dynamics (motion of individual atoms and molecules).
Maxwell and Boltzmann used the model to derive the distribution of speeds of
interacting molecules as a function of temperature;
Einstein used it to explain the Brownian motion of pollen grains immersed in water.
Simulation.
There are two natural approaches to simulating the system of
particles.
Collision prediction.
In this section we present the formulas that specify if and
when a particle will collide with the boundary or with another particle,
assuming all particles travel in straight-line trajectories.
- Collision between particle and a wall.
Given the position (rx, ry), velocity
(vx, vy), and radius σ of
a particle at time t, we wish to determine if and when it
will collide with a vertical or horizontal wall.
Since the coordinates are between 0 and 1, a particle comes into
contact with a horizontal wall at time t + Δt if
the quantity ry + Δt vy equals either
σ or (1 - σ). Solving for Δt yields:
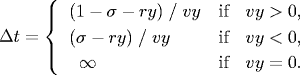
An analogous equation predicts the time of collision with a vertical wall.
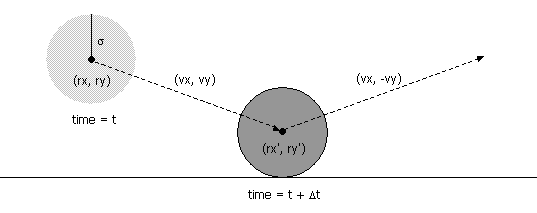
- Collision between two particles.
Given the positions and velocities of two particles i and j
at time t,
we wish to determine if and when they will collide with each other.
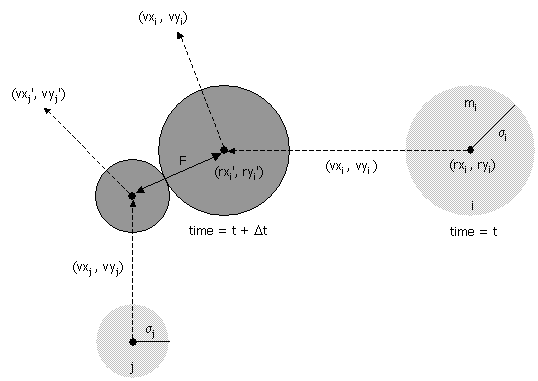
Let (rxi' , ryi' ) and
(rxj' , ryj' ) denote the positions
of particles i and j at the moment of contact, say
t + Δt.
When the particles collide, their
centers are separated by a distance of σ = σi + σj.
In other words:
σ2 =
(rxi' - rxj' )2 + (ryi' - ryj' )2
During the time prior to the collision, the particles move in
straight-line trajectories. Thus,
rxi' = rxi + Δt vxi, ryi' = ryi + Δt vyi
rxj' = rxj + Δt vxj, ryj' = ryj + Δt vyj
Substituting these four equations into the previous one, solving the
resulting quadratic equation for Δt, selecting the
physically relevant root,
and simplifying, we obtain an expression for Δt
in terms of the known positions, velocities, and radii.

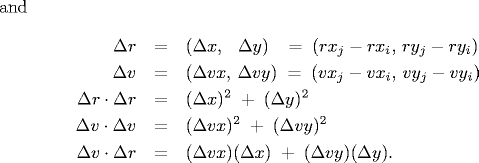
If either Δv ⋅ Δr ≥ 0
or d < 0, then the quadratic equation has no solution for Δt > 0;
otherwise we are guaranteed that Δt ≥ 0.
Collision resolution.
In this section we present the physical formulas that specify the
behavior of a particle after an elastic collision with a reflecting
boundary or with another particle.
For simplicity, we ignore multi-particle collisions.
There are three equations governing
the elastic collision between a pair of hard discs:
(i) conservation of linear momentum, (ii) conservation of kinetic energy,
and (iii) upon collision, the normal force acts perpendicular to the
surface at the collision point.
Physics-ly inclined students are encouraged to derive the equations
from first principles; the rest of you may keep reading.
- Between particle and wall.
If a particle with velocity (vx, vy)
collides with a wall perpendicular to x-axis, then the
new velocity is (-vx, vy); if it collides with a wall
perpendicular to the y-axis, then the
new velocity is (vx, -vy).
- Between two particles.
When two hard discs collide, the normal force acts along
the line connecting their centers (assuming no friction or spin).
The impulse (Jx, Jy) due to the normal force in the
x and y directions of a perfectly elastic collision
at the moment of contact is:
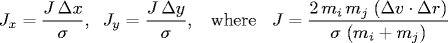
and where mi and mj are the masses of
particles i and j, and
σ, Δx, Δy and Δ v ⋅ Δr
are defined as above.
Once we know the impulse, we can apply
Newton's second law (in momentum form) to compute the velocities immediately after
the collision.
vxi' = vxi + Jx / mi, vxj' = vxj - Jx / mj
vyi' = vyi + Jy / mi, vyj' = vyj - Jy / mj
Data format.
Use the following data format.
The first line contains the number of particles N.
Each of the remaining N lines consists of 6 real numbers
(position, velocity, mass, and radius) followed by
three integers (red, green, and blue values of color).
You may assume that all of the position coordinates are
between 0 and 1, and the color values are between 0 and 255.
Also, you may assume that none of the particles intersect
each other or the walls.
N
rx ry vx vy mass radius r g b
rx ry vx vy mass radius r g b
rx ry vx vy mass radius r g b
rx ry vx vy mass radius r g b
Your task.
Write a program MDSimulation.java that reads in a set of
particles from standard input and animates their motion according to the
laws of elastic collisions and event-driven simulation.
Perhaps everything below is just for the checklist??
Particle collision simulation in Java.
There are a number of ways to design a particle collision simulation program
using the physics formulas above.
We will describe one such approach, but you are free to substitute
your own ideas instead.
Our approach involves the following
data types: MinPQ, Particle, CollisionSystem,
and Event.
Data sets.
Some possibilities that we'll supply.
- One particle in motion.
- Two particles in head on collision.
- Two particles, one at rest, colliding at an angle.
- Some good examples for testing and debugging.
- One red particle in motion, N blue particles at rest.
- N particles on a lattice with random initial directions
(but same speed) so that the total kinetic energy is consistent with
some fixed temperature T, and total linear momentum = 0.
Have a different data set for different values of T.
- Diffusion I: assign N very tiny particles of the same size
near the center of the container with random velocities.
- Diffusion II: N blue particles on left, N green particles on right
assigned velocities so that they are thermalized (e.g., leave
partition between them and save positions and velocities after
a while). Watch them mix. Calculate average diffusion rate?
- N big particles so there isn't much room to move without
hitting something.
- Einstein's explanation of Brownian motion:
1 big red particle in the center, N smaller blue particles.
Things you could compute.
- Brownian motion.
In 1827, the botanist Robert Brown observed
the motion of wildflower pollen grains immersed in water
using a microscope.
He observed that the pollen grains were in a random
motion, following what would become known as Brownian motion.
This phenomenon was discussed, but no convincing explanation
was provided until Einstein provided a mathematical one in 1905.
Einstein's explanation: the motion of the pollen grain particles
was caused by millions of tiny molecules colliding with the
larger particles. He gave detailed formulas describing the
behavior of tiny particles suspended in a liquid at
a given temperature.
Einstein's explanation was intended to help justify the
existence of atoms and molecules and could be used to
estimate the size of the molecules.
Einstein's theory of Brownian motion enables engineers to
compute the diffusion constants of alloys by observing
the motion of individual particles.
Here's a flash demo of
Einstein's explanation of Brownian
motion from
here.
- Free path and free time.
Free path = distance a particle travels between collisions.
Plot histogram. Mean free path = average free path length over
all particles.
As temperature increases, mean free path increases (holding pressure constant).
Compute free time length = time elapsed before a particle
collides with another particle or wall.
- Collision frequency.
Number of collisions per second.
- Root mean-square velocity.
Root mean-square velocity / mean free path = collision frequency.
Root mean square velocity = sqrt(3RT/M) where
molar gas constant R = 8.314 J / mol K,
T = temperature (e.g., 298.15 K), M = molecular mass
(e.g., 0.0319998 kg for oxygen).
- Maxwell-Boltzmann distribution.
Distribution of velocity of particles in hard sphere model
obey Maxwell-Boltzmann distribution (assuming system has thermalized
and particles are sufficiently heavy that we can discount quantum-mechanical
effects). Distribution shape depends on temperature.
Velocity of each component has distribution proportional to
exp(-mv_x^2 / 2kT). Magnitude of velocity in d dimensions
has distribution proportional to v^(d-1) exp(-mv^2 / 2kT).
Used in statistical mechanics because it is unwieldy to simulate
on the order of 10^23 particles.
Reason: velocity in x, y, and z directions are normal (if
all particles have same mass and radius).
In 2d, Rayleigh instead of Maxwell-Boltzmann.
- Pressure.
Main thermodynamic property of interest = mean pressure.
Pressure = force per unit area exerted against container by
colliding molecules.
In 2d, pressure = average force per unit length on the wall of
the container.
- Temperature.
Plot temperature over time (should be constant) = 1/N sum(mv^2) / (d k),
where d = dimension = 2, k = Boltzmann's constant.
- Diffusion.
Molecules travel very quickly (faster than a speeding jet)
but diffuse slowly because they collide with other molecules,
thereby changing their direction.
Two vessels connected by a pipe containing two different types
of particles. Measure fraction of particles of each type in
each vessel as a function of time.
- Time reversibility.
Change all velocities and run system backwards.
Neglecting roundoff error, the system will return to its original
state!
- Maxwell's demon.
Maxwell's demon
is a thought experiment conjured up by
James Clerk Maxwell in 1871 to contradict the second law of thermodynamics.
Vertical wall in middle with molecule size trap door,
N particles on left half and N on right half,
particle can only go through trap door if demon lets it through.
Demon lets through faster than average particles from left to right,
and slower than average particles from right to left.
Can use redistribution of energy to run a heat engine by allowing
heat to flow from left to right.
(Doesn't violate law because demon must interact with the physical
world to observe the molecules. Demon must store information about
which side of the trap door the molecule is on.
Demon eventually runs out of storage space and must begin erasing
previous accumulated information to make room for new information.
This erasing of information
increases the entropy, requiring kT ln 2 units of work.)
Cell method.
Useful optimization: divide region into rectangular cells.
Ensure that particles can only collide with particles in one of
9 adjacent cells in any time quantum. Reduces number of binary collision
events that must be calculated. Downside: must monitor particles
as they move from cell to cell.
Extra credit.
Handle multi-particle collisions. Such collisions are important when
simulating the break in a game of billiards.